 
For example, as we move down a group, the metallic character of the atoms increases. Semimetals exhibit properties intermediate between those of metals and nonmetals. However, there are also other patterns in chemical properties on the periodic table. There is a recurring pattern called the periodic law in their properties, in which elements in the same column (group) have similar properties. It is organized in order of increasing atomic number. The periodic law is the principle that the properties or characteristics of the elements recur. The periodic table is a tabular arrangement of the chemical elements. The elements can be broadly divided into metals, nonmetals, and semimetals. A period is a horizontal row of the periodic table. Some of the groups have widely-used common names, including the alkali metals (Group 1) and the alkaline earth metals (Group 2) on the far left, and the halogens (Group 17) and the noble gases (Group 18) on the far right. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods. 
It arranges of the elements in order of increasing atomic number. The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. 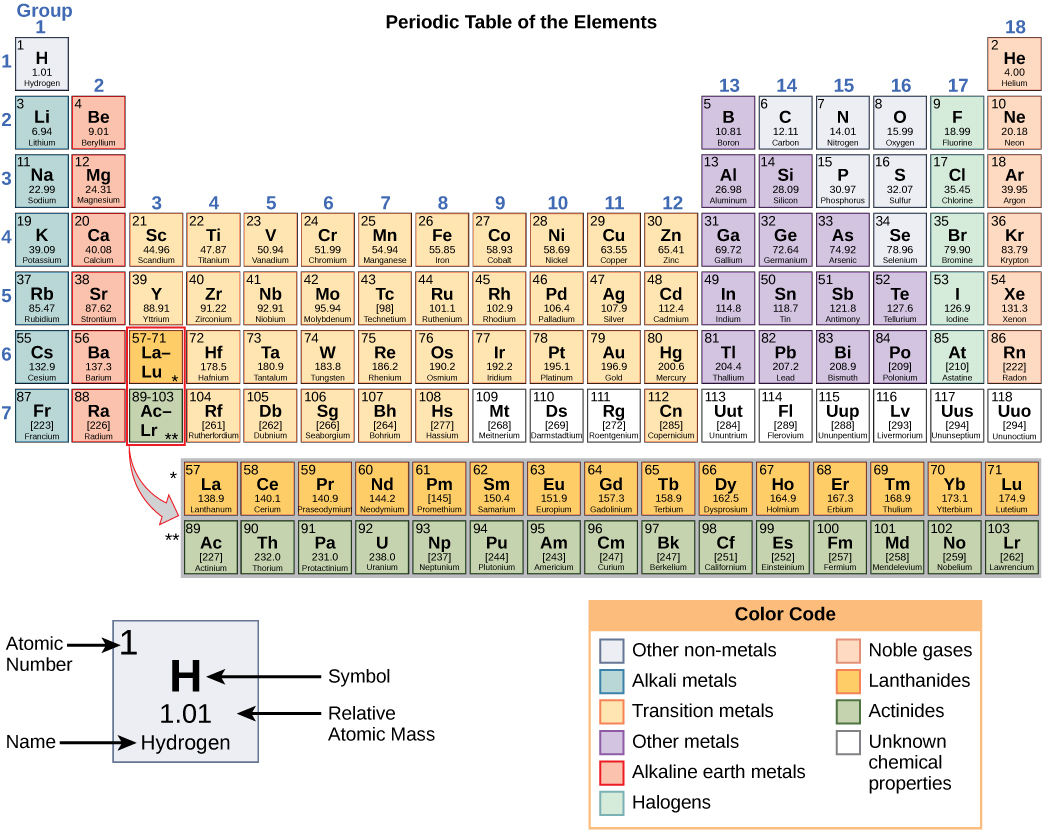
Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. The simplest definition is that a period begins when a new s-subshell starts to fill.Recall that in the s, p, d, f subshells there are 2, 8, 10, and 14 electrons, respectively, so the periods have to get bigger over time and thus cannot be regular. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. I agree that the 'periods' in the periodic table are not mathematically regular. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns. Periodic trends, arising from the arrangement of the periodic table, provide chemists with an invaluable tool to quickly predict an elements properties. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
